The initial Egaceutical study was unpublished and observational. Each study subject paid for their involvement in the study, was under the care of their own physician, and then provided their results to us. We did not verify the results they presented to us. The study was not peer reviewed or double blinded.
The study showed reversal of biological age with triple-compound EGA® in 12 human males from age 46 to 66 with no negative side effects.
The original 12 were a group of friends in what was originally intended to be just an experiential experiment with NMN to see if it was true what David Sinclair, of Harvard Medical School, was claiming in the world press. There was not a desire for a company or profit at the time, just a group who did not want to grow old and were willing to take a chance.
To lower group risk, a single individual went first, so the rest of the group waited for over a year to start. It was not prudent for the whole group to face any risk until a single individual had taken the NMN for the 8 months which was estimated to be the equivalent of the single week the mice were on the NMN to get the Harvard Medical School result. Then the next 3 men started on EGA®, then another 8.
Egaceutical Corporation is now starting the 4th wave of individuals on EGA®. It is possible to join this group. To join, see the section on Present Studies.
The experience of the first individual on EGA® has been written up in the patent application; see that section of this website on page 88 of the patent application for that account. A video at the end of this section also details his experience.
To summarize, after three months on the NMN alone, the effects of NMN wore off and we looked into the scientific literature to try to understand why. It was realized then that sirtuin enzymes are pattern recognition enzymes and are only turned on if they see the correct pattern of events in the cell. There are several ways to turn the sirtuin enzymes off. The solution was to add two other ingredients to stop this turning off of the sirtuin enzymes. The additional two ingredients not only stopped the effect of the singular compound NMN from going away but increased the effect of the therapy. This realization led to the patent application and to the formation of the company. All subsequent individuals in the initial study only took the triple compound EGA®, not the singular NMN. The patent application was submitted before the next group of three and the subsequent group of 8 started on EGA® so their results were not included in the patent application.
Results from the initial study of 12 males
First and most importantly: none of the 12 had reported to us any negative side effects during the time they were on EGA®. The main objective of the study was accomplished: all that were measured lowered their IL-6 and TNF-α, the key endpoints in the preliminary study. The reasons for choosing Interleukin-6 (IL-6) as the primary end point are discussed below. Wonderful secondary results were reported by our healthy study members consistent with a more youthful physicality and mentality, The participants self-reported: improved energy, improved sleep, more mental alertness, improved cognition (e.g. better chess rating), improved physical endurance, increased strength, increased muscle mass, decreased body fat, weight loss, increased joint mobility and flexibility, elimination of body aches and pains, elimination of arthritis in the 3 out of 12 males that had arthritis, better vision at eye exam , healthier skin, and disappearance of aged appearance of skin cells.
First person (Joel) on EGA® talks about the experience
Second person (Lawrence) on EGA® talks about the experience
IL-6 as a prediction for the “diseases of aging”
For mitochondrial health → Correia-Melo C 2016
For immune and auto-immune disease → Astrakhantseva IV 2004
For heart disease → Ridker P 2000
For stroke → Herder C 2011
For kidney disease → Lee B 2015
For Alzheimer’s disease → Butchart J 2015 & Holmes C 2011
For defense against bacteria → Puchta A 2016
For defense against viruses → McElroy AK 2016
For intelligence → Bettcher B 2014 (multiple) 2015
For sleep disorders → Irwin MR 2015
For physical performance → Cesari M 2004
IL-6 as a prediction for longevity and all-cause mortality
Puzianowska-Kuznicka M 2016 & Arai Y 2015 & Varadhan R 2014 & Lee JK 2012 & Derhovanessian E 2010 & Bruunsgaard H 2003 & Reuben DB 2002 & Taaffe DR 2000
Also grouped with the IL-6 test in Arai Y 2015 are
Tumor necrosis factor Alpha (TNF-α)
hsCRP
CMV-IgG
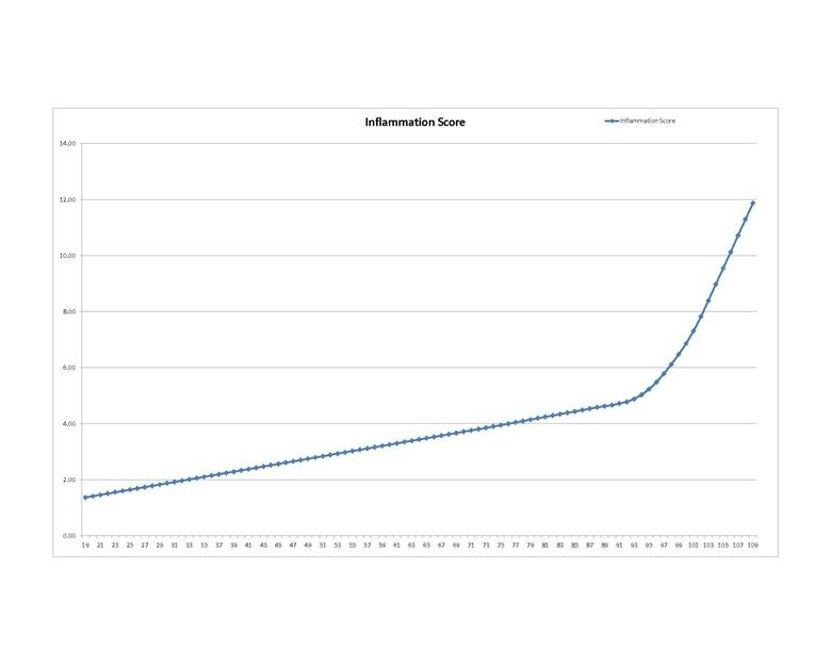
Comparisons (of testing before the start and monthly tests while taking EGA®) were made to this graph generated (extrapolated in the low/youth range) from data using the above 4 inflammation tests and results from data of same tests described in Arai Y 2015.